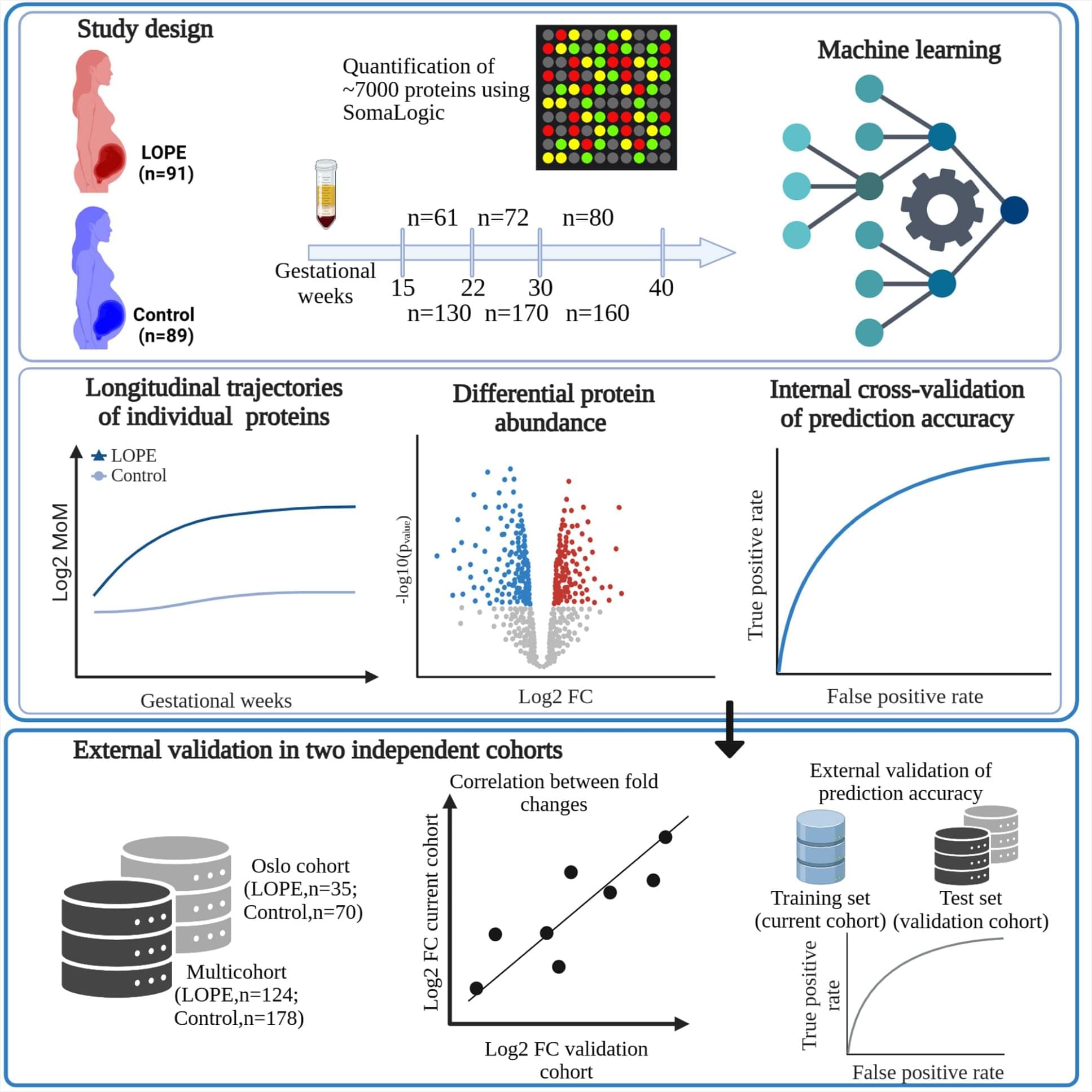
The future of intelligence is rapidly evolving with AI advancements, poised to transform numerous aspects of life, work, and existence, with exponential growth and sweeping changes expected in the near future.
## Questions to inspire discussion.
Strategic Investment & Career Focus.
🎯 Q: Which companies should I prioritize for investment or career opportunities in the AI era?
A: Focus on companies with the strongest AI models and those advancing energy abundance, as these will have the largest marginal impact on enabling the innermost loop of robots building fabs, chips, and AI data centers to accelerate exponentially.
Understanding Market Dynamics.