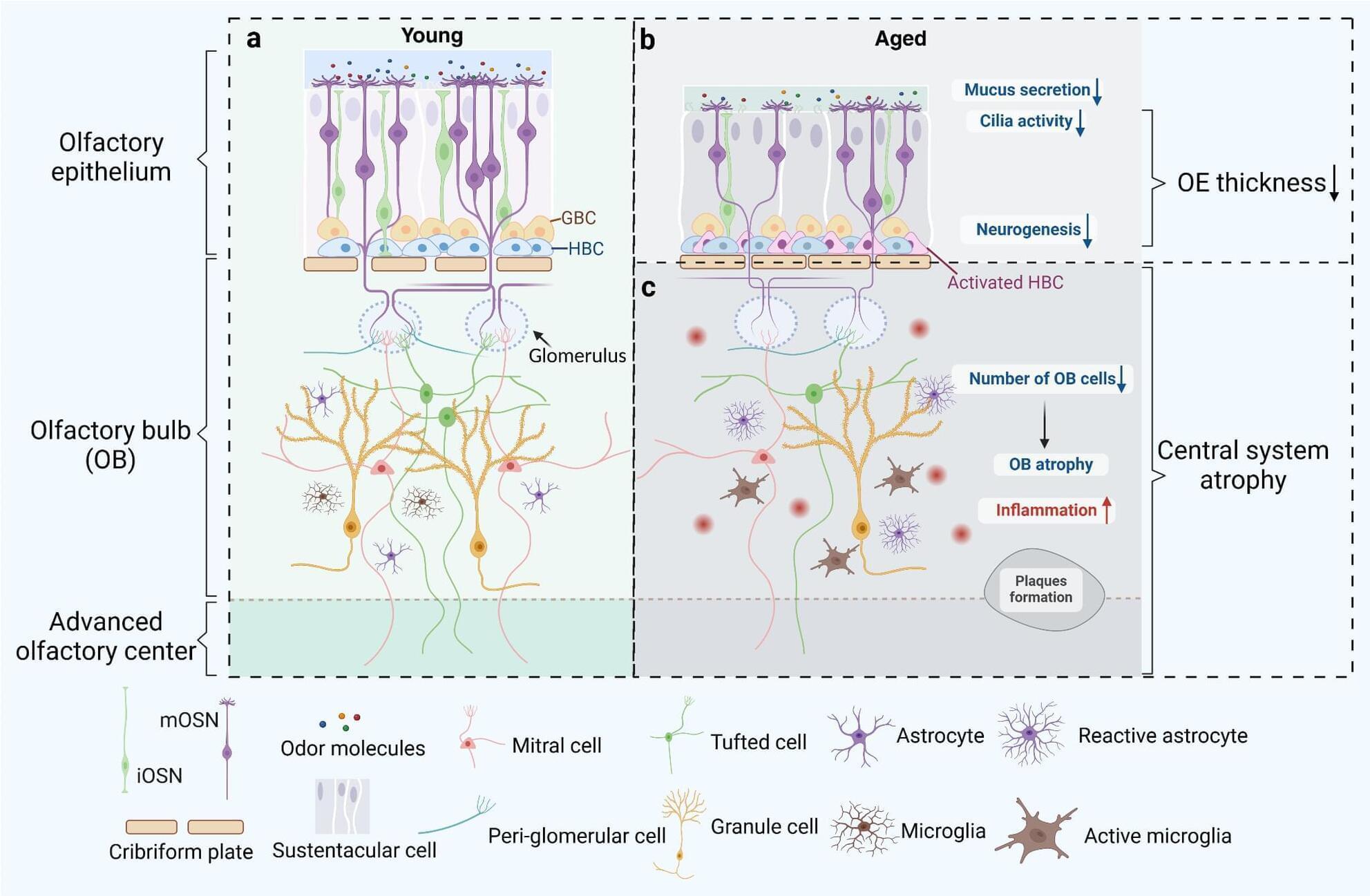
Olfactory dysfunction (OD) is a common nasal disease, particularly prevalent among the elderly population, significantly impacting the affected individuals’ quality of life. This review focuses on the influence of aging and chronic inflammation on olfactory dysfunction, presenting insights from both the peripheral and central olfactory systems. By exploring the molecular mechanisms and pathological changes underlying the occurrence of olfactory dysfunction in relation to age-related diseases and chronic inflammation conditions, we aim to provide a comprehensive theoretical foundation for further research and offer valuable insights for more effective treatment of olfactory dysfunction.