Chemists made a photocatalytic discovery when their control reaction led to an unexpected product





Despite this reality, an excessive number of organizations, notably small and mid-sized businesses (SMBs), lack designated leadership in this crucial domain.
#cybersecurity #business #ciso #tech
By Chuck Brooks.
Cybersecurity is no longer an IT concern; it has become a fundamental business requirement. Viability, survivability, and ultimately commercial success are dependent on securing the devices and network for any business in the evolving digital era.
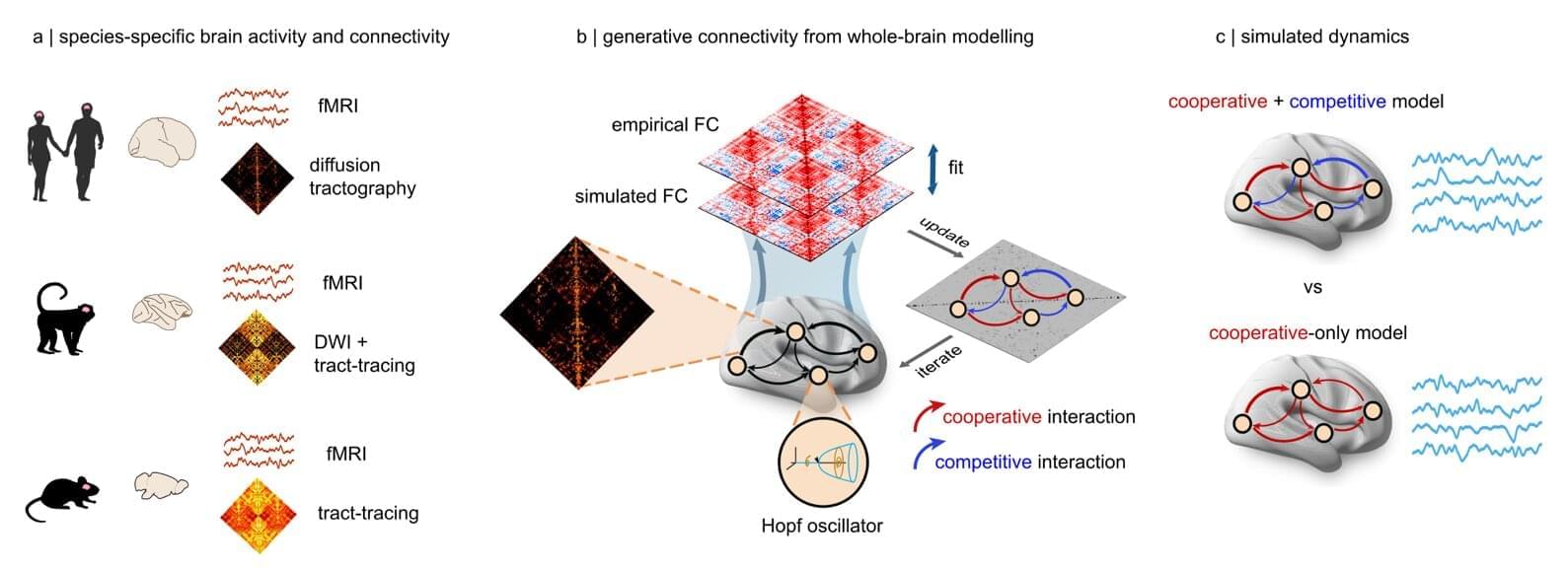
Over the past decades, neuroscience studies have painted an increasingly detailed picture of the human brain, its organization and how it supports various functions. To plan and execute desired behaviors in changing circumstances, networks of neurons in the brain can either work together or suppress each other, thus employing both cooperative and competitive interaction strategies.
Researchers at University of Oxford, University of Cambridge, McGill University, University of Aarhus and Pompeu Fabra University recently set out to better understand the mammalian brain’s underlying dynamics, specifically how its underlying architecture balances cooperative and competitive interactions between neural circuits. Their paper, published in Nature Neuroscience, offers new insight that could both improve the understanding of the brain and inform the development of brain-inspired computational models.
“Building models of the brain is an important part of modern neuroscience,” Andrea Luppi, first author of the paper, told Medical Xpress. “As Nobel winner Reichard Feynman said, ‘what I cannot create, I do not understand.’ Most current models, however, share a limitation. Everyday experience, from focusing attention or switching between tasks, also reveals that brain systems must compete for limited resources.

Northwestern Medicine scientists led by Joseph Bass, MD, Ph.D., the Charles F. Kettering Professor of Endocrinology and Metabolism and director of the Center for Diabetes and Metabolism, have discovered how disruptions in the circadian rhythm impair metabolic function in fat cells, providing new insights into the molecular mechanisms that cause obesity and metabolic disease, according to a recent study published in Nature Metabolism.
“It’s not simply the accrual of excess fat that leads to disease. It’s a change in the actual function and the capacity of the energy center within the cell to work properly,” said Bass, who is also chief of Endocrinology, Metabolism and Molecular Medicine in the Department of Medicine and a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
The circadian rhythm is the body’s own internal 24-hour clock that regulates the sleep-wake cycle, hormone levels and metabolism, among other systems throughout the body.

(SRI) will organize a high-level side event during the COPUOS Legal Subcommittee on 16 April 2026 at UNOOSA (Vienna), proposed and convened by Dr. Gülin Dede, titled “Operationalising Space as a Cross-Cutting Enabler of Sustainable Development: Perspectives on an Emerging 18th SDG Articulation.”
The session will bring together legal, policy, industry, and Global South perspectives to examine how outer space is evolving from a sectoral domain into a critical enabling infrastructure for the 2030 Agenda, while simultaneously requiring stewardship as an environment in its own right.
Positioned as an early contribution to shaping how space sustainability is framed within the broader UN system, the event will also be broadcast by the United Nations, extending its reach beyond the room to a global audience.